

BellaSeno Reports Positive Interim Findings of Australian Clinical Trials in Breast Scaffold Patients

Funding to support establishment of the world´s first fully automated production facility for resorbable medical scaffolds based in Australia

BellaSeno Appoints Dr. Thomas Lingner as Chief Technology Officer and Inga Freyert as VP Quality and Regulatory Affairs

BellaSeno’s 3D-printed Resorbable Scaffold
Successfully Used in Critical-Size Large Segmental Radius Bone Defect

New investigator-initiated clinical trials
BellaSeno’s resorbable tissue regeneration scaffolds to be conducted in Europe
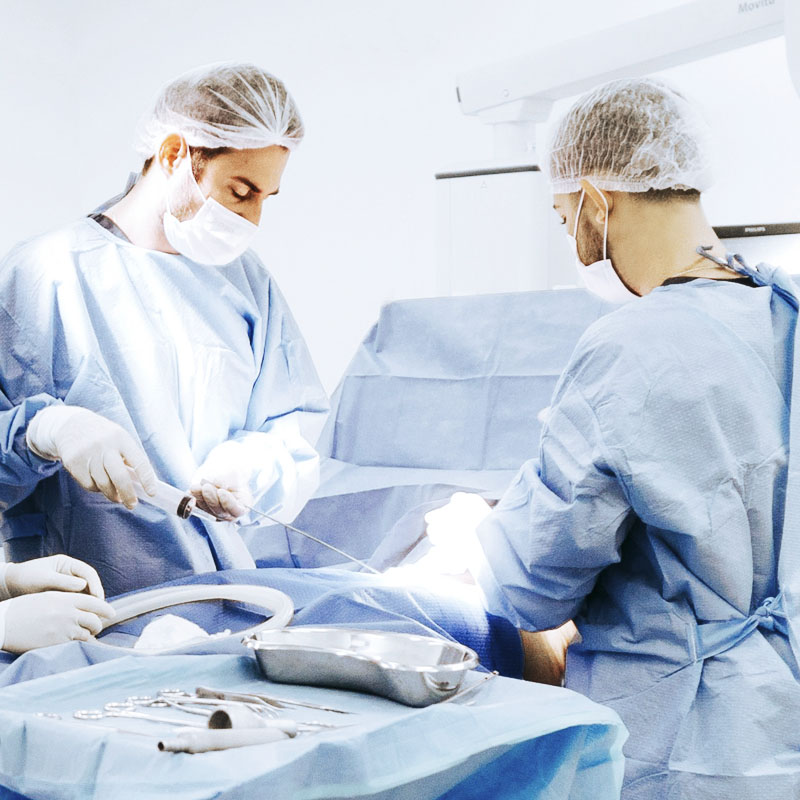
BellaSeno Presents Novel Production Workflow
Production Workflow for Customized, 3D-Printed Bone Scaffolds
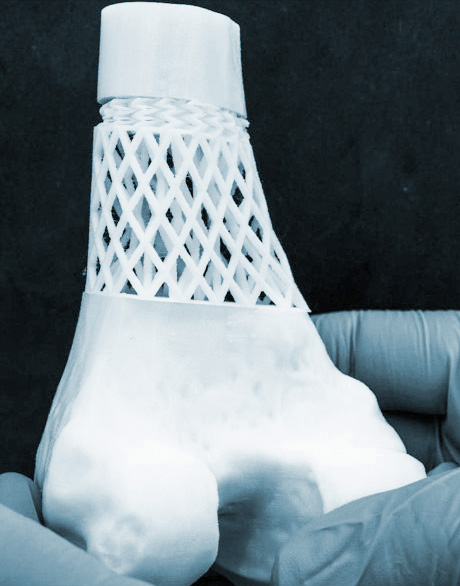
Positive Follow-Up Data
BellaSeno Reports Positive One-Year Patient Follow-Up Data for Resorbable Breast and Pectus Excavatum Scaffolds
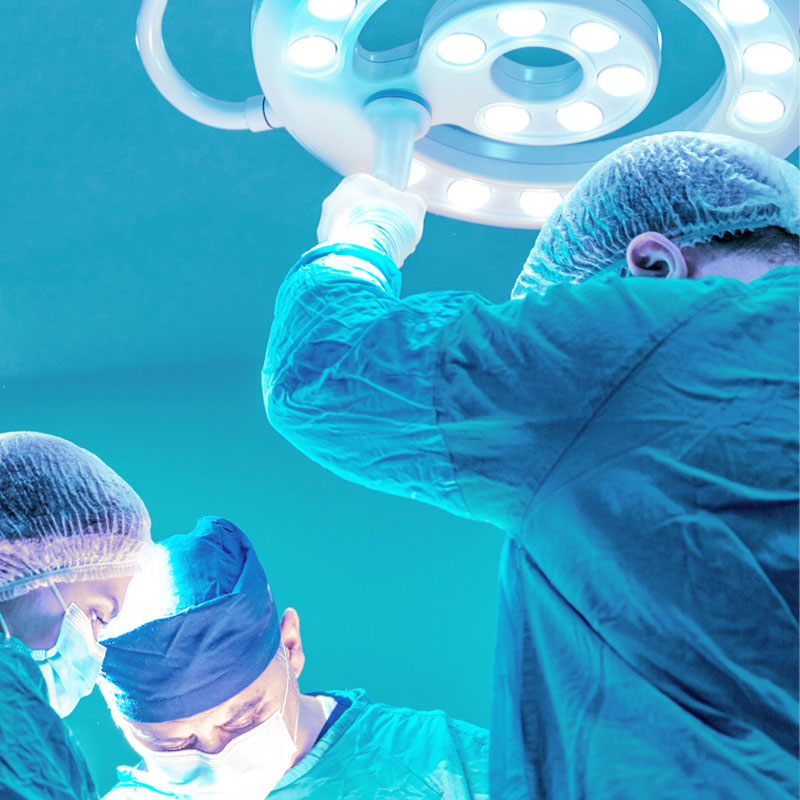
Granted Market Access
BellaSeno Granted Market Access in Europe for Custom-Made Bone and Pectus Excavatum Scaffolds

BellaSeno Demonstrates Superior Biomechanical Properties of its 3-D Printed, Resorbable Scaffolds for Bone Reconstruction

Positive Interim Data of BellaSeno’s Clinical Trial with Resorbable Breast and Chest Implants Reported at RACS 2023

BellaSeno Starts Two Clinical Trials of its Resorbable Breast and Chest Implants
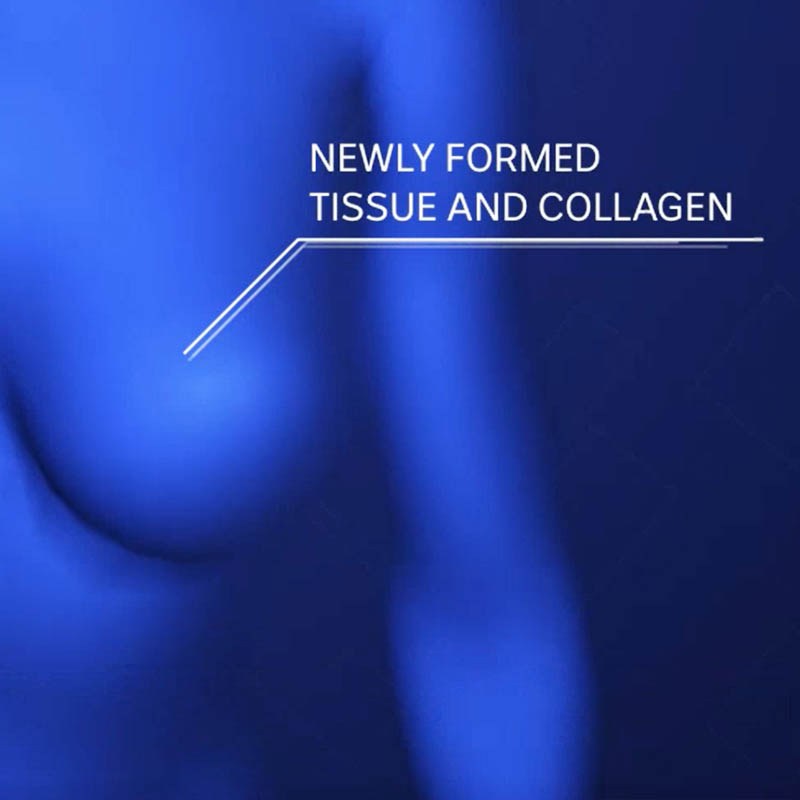
Pectus excavatum camouflage
The first-in-human trial of soft tissue engineering to camouflage a pectus excavatum defect not reconstructable by conventional techniques.
